The company PharmaInformatic is specialized in the development of expert systems. Current scientific information from computer science (cheminformatics, bioinformatics and artificial intelligence) are used to specifically develop software products and services for the pharmaceutical industry. Computer programs can be used to reduce animal use; according to the manufacturer, predictions made by these expert systems are more precise and can estimate the oral bioavailability in humans more exactly and reliably than in animal experiments. Furthermore, potential drug candidates can be recognized before testing, thus reducing development costs.
In the sign of the molecule: The PharmaInformatic logo.
Source: PharmaInformatic.
The company focuses on the development of an expert system to assess the bioavailability of a substance in the body. An expert system is a computer program that helps the user to answer complex questions as a specialist would. In the process it gives recommendations for action on the basis of a knowledge base. This system, called IMPACT-R, obtains its data bases from a dedicated knowledge base, PACT-F.
PACT-F stands for “Preclinical And Clinical Trials Knowledge Base on Bioavailability”.
Manufacturers are legally required to carry out both preclinical trials and clinical trials during the development of new drugs. In preclinical trials, animals are used if no accepted animal-free replacement methods are available. Clinical trials are subsequently conducted if the preclinical tests show the test substance to be neither dangerous nor disadvantageous to the test organism.
What is bioavailability?
Bioavailability is an important part of the toxicokinetic process, the so-called ADME properties of a substance:
A absorption
D distribution
M metabolism
E excretion
“Bioavailability” describes A = absorption and M = metabolism.
The term “bioavailability” means the availability of substances administered extravascularly (outside the blood or lymph vessels) for its effect, i.e. the portion that actually reaches the systemic circulation and thus the site of action1. This implies that in order to transport a substance to the target organ, first it must be absorbed through the stomach or intestinal mucosa. From there it enters the bloodstream, is bound to transport proteins and transported. If a substance is taken orally, its amount is usually reduced by an incomplete absorption via the gastric and intestinal mucosa and a so-called pre-systemic metabolism in the intestinal mucosa and liver. The latter process is called the “first-pass effect”.
The knowledge of the extent of these processes is extremely important in order to determine the appropriate substance dose.
The bioavailability of an intravenously administered substance is initially 100 per cent in the blood plasma, then the concentration decreases in a specific curve (see below) because the substance is biotransformed and excreted. Conversely, extravascularly (e. g. orally) administered substances only enter the blood plasma after a certain time with a different the concentration gradient.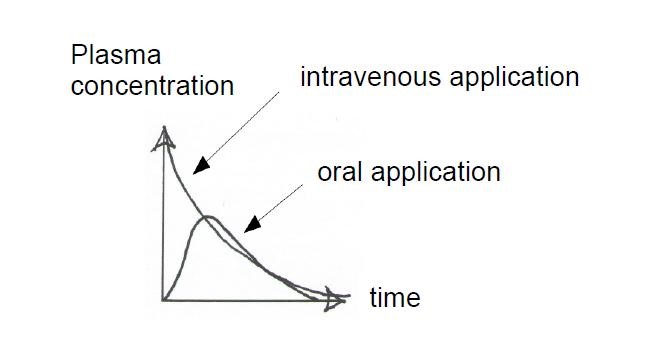
The bioavailability is depends greatly on the first-pass effect: Once the substance has been absorbed by the stomach and intestinal mucosa, it passes the portal vein into the liver where it is metabolized for the first time. Part of it destined for excretion and does not reach the target organ. The rest passes through the hepatic vein back into the bloodstream where it is transported to the target organ, and is thus bioavailable. All these processes differ both between humans and animals, and animals of different species. They also differ in some extent between individual humans1 (“Inter-subject variability” (see also: http://www.pharmainformatic.com/ html / prediction_of_f_.html # Inter-subject variability).
PACT F
The knowledge base PACT F contains nearly 8,300 entries with results from preclinical and clinical studies of bioavailability. Each entry contains the chemical structure of the substance under study, as well as other information, such as substance formulation, the route of administration (i.e., how the substance is taken: orally, subcutaneously, intravenously, intramuscularly, intraperitoneally, or rectally)2.
Source: PharmaInformatic.
The metabolic classes involved in the conversion of the substance in the body and preparing for excretion play a very large role. Here especially metabolic enzymes are of great importance, which are present in various human genetic polymorphisms, i.e. the gene is present multiple times, due to resulting in accelerated degradation, so that it is less effective or not at all. However, there are also other mechanisms by which metabolic enzymes interact and can cause side effects. All these mechanisms be considered and evaluated before the test procedure commences.
Other important database information is data on dosage frequency, in the case of clinical studies on humans, age, sex, and ethnicity. The health status of the study participants is also recorded and is an important factor for the uptake of a substance into the bloodstream. Participants with impaired liver function can achieve a much higher bioavailability than healthy subjects.
In the case of preclinical studies, details concerning the animal species used, health status and feeding conditions are relevant.
IMPACT F
The expert system IMPACT-F predicts the oral bioavailability of a substance in humans. The calculations are based on reliable computer models3.
The performance and quality of the models were checked by 34 different pharmaceutical test candidates intended for use in cancer therapy, as anti-infective agents and for treating disorders of the central nervous system, the gastrointestinal tract and the kidneys, and inflammatory processes.
Source: PharmaInformatic.
The model’s predictions were more accurate than preclinical tests on animals. The predictions correlated well with the experimental results from clinical drug tests.
A licence is necessary to use the expert system.
Species differences in bioavailability after oral administration
As part of the development of the expert system, the differences between the species were also examined in more detail4.
Analysis of the knowledge base PACT-F allowed a comparison of the experimental results of animal studies and human clinical trials based on more than a hundred approved drugs.
The research results (see bar graph) show that the oral bioavailability measured in animals did not match the value determined in humans for a variety of drugs: Some of the differences in oral bioavailability between humans and animals were considerable.
For example, the knowledge base evaluation showed large differences in the oral bioavailability of the antibiotic ciprofloxacin between humans and animals.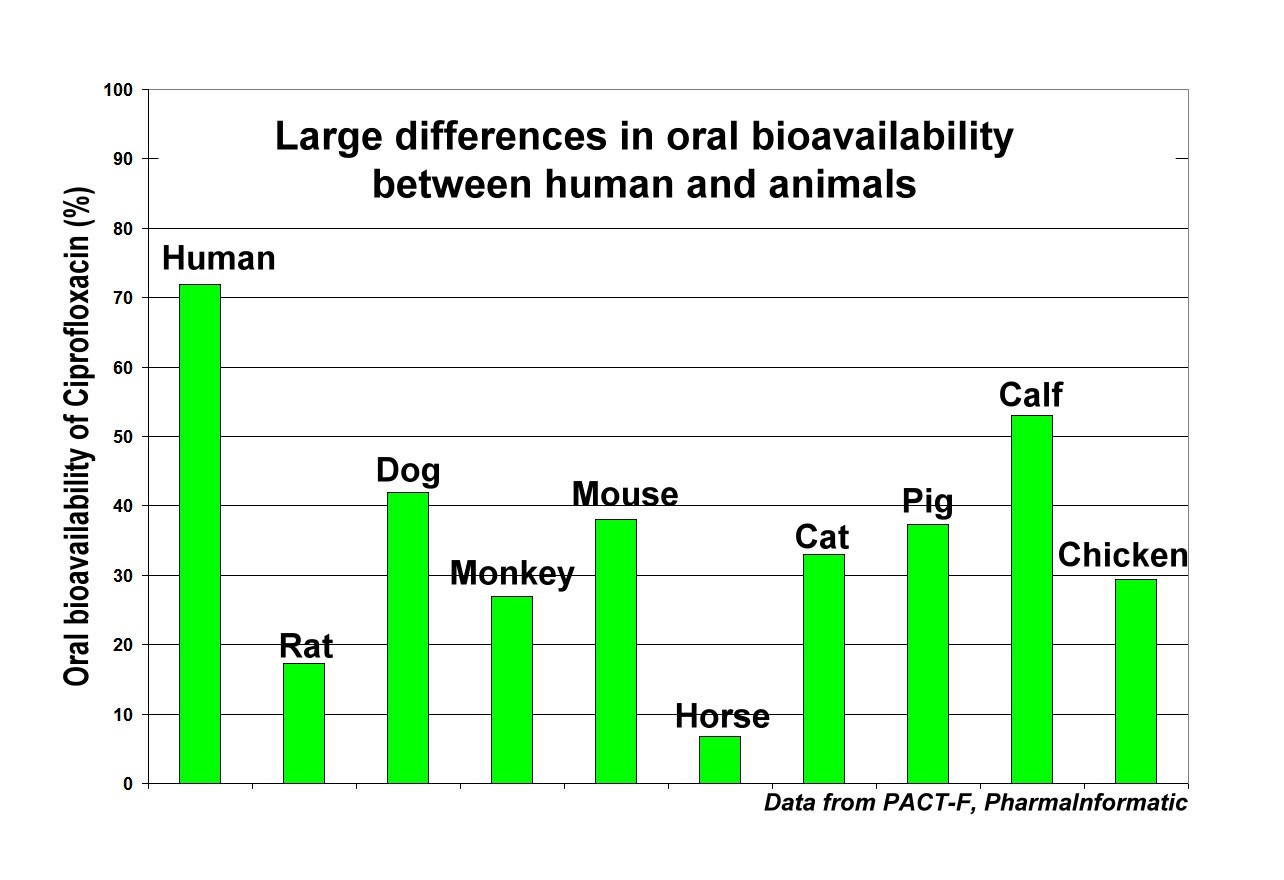
Graphics: BioInformatic.
In addition to the precision of the approach, a further advantage of this expert system is the capacity for saving animals where they are used to determine the dose of substance.
“... significantly more accurate and reliable than animal testing ...”
InVitroJobs interviewed the founder and director of PharmaInformatic, Dr. Wolfgang Boomgaarden about his expert systems.
Image: Wolfgang Boomgaarden.
InVitroJobs: How did you come up with the idea of developing a database for the bioavailability of drugs?
Dr. Boomgaarden: Years ago, in discussions with research-based pharmaceutical companies, I was asked whether in addition to our other products we were also able to predict the oral bioavailability of new drugs.
Oral bioavailability is one of the most important characteristics when developing new drugs. In order to develop such prediction models, one needs a very large amount of data and information, which must be correlated to the respective substances and chemical structural formulas.
The development of the knowledge database PACT-F took many years, as the detailed results and experimental conditions of more than 5,000 scientific publications on the bioavailability of active ingredients had to be evaluated manually, analyzed, and structured.
Although this development process was very labour-intensive and arduous, it was worth it: We now have the world’s largest and most annotated knowledge database on the bioavailability of drugs and medicines, which is used for general research into the bioavailability of drugs.
InVitroJobs: What have you investigated and what lessons have you drawn?
Dr. Boomgaarden: So far we have examined in detail how suitable animal tests are for predicting the oral bioavailability of drugs in humans.
In today’s pharmaceutical research, many new drug candidates are tested on animals in order to determine whether these agents have sufficient oral bioavailability in those animals.
But what drug candidate is most suitable for further development to a new drug?
The oral bioavailability measured in animals may be used as a criterion for deciding whether further clinical studies are carried out.
For the first time we have now been able, on the basis of a very large number of approved drugs, to determine and quantify to what extent oral bioavailability in animals and humans correspond and correlates.
Our results demonstrate that the oral bioavailability measured in animals for a large number of drugs does not correspond to value with the determined.
We have found considerable differences for in oral bioavailability between humans and animals for many drugs and have published our findings on our websites (http://www.pharmainformatic.com/html/prediction_of_f_.html).
We have also used the knowledge database to develop the expert system IMPACT-F, which determines the oral bioavailability in humans of prospective active agents and medicines.
In the process, we found out that the expert system can estimate the oral bioavailability in humans far more accurately and reliably than bioavailability studies using animals.
InVitroJobs: For which scientific questions can the knowledge database PACT-F be used?
Dr. Boomgaarden: The knowledge base can be used to help research the molecular causes, reasons and factors that influence why and to what extent substances are bioavailable in animals or in humans.
The knowledge database is extensively annotated. This means that a large number of parameters and factors that may affect the bioavailability were integrated into the knowledge database.
Here are some examples:
Are there gender-specific or genetic factors?
What effect does a co-administration of other drugs have?
What diseases influence bioavailability?
What effect does the subject’s age have?
Does food intake affect oral bioavailability?
Which substance or metabolite was analyzed?
Which measurement method is used?
What does this method imply and were reliable data generated?
The knowledge database forms the fundamental basis to develop new theories, contexts and rules on the bioavailability of drugs and to optimize the oral bioavailability of future drugs.
InVitroJobs: What are the advantages of the expert system IMPACT-F compared to animals in the research and development of new drugs?
Dr. Boomgaarden: The expert system IMPACT-F means that drug candidates can now be evaluated for oral bioavailability in a very early phase of drug development. This allows the selection of appropriate development candidates and the elimination of unsuitable candidates at an early stage. This is something the pharmaceutical industry has been demanding for a long time as a way of reducing the considerable costs of drug development.
As opposed to animal experiments, the results are available directly and more cost-effective. No further costs are incurred, such as by chemical synthesis of active agents.
Crucial, however, is the quality: The expert system can significantly predict the oral bioavailability in humans significantly more accurately and reliably than animal tests can: The accuracy of the predictions made by IMPACT-F corresponded to the deviation typically measured between individual human in clinical trials.
InVitroJobs: How has industry responded?
Dr. Boomgaarden: Very encouragingly! Several pharmaceutical companies already use IMPACT-F for predicting the oral bioavailability of their research candidates, and they do this in various stages of drug development: on the one hand during the preclinical phase, where suitable research candidates are optimized and selected, on the other hand during preparation and support of human clinical studies.
And all this just two weeks after launching the expert system onto the market! I think this also shows the great interest of pharmaceutical companies in this expert system.
InVitroJobs: Thank you very much for the interview.
Literature:
1 Aktories et al. (2009): Repetitorium Allgemeine und spezielle Pharmakologie und Toxikologie. Munich.
2 Knowledge Base PACT-F. http://www.pharmainformatic.com/html/databases.html
3 Expert system IMPACT-F. http://www.pharmainformatic.com/html/impact-f.html
4 Research Results: http://www.pharmainformatic.com/html/prediction_of_f_.html
und
http://www.pharmainformatic.com/html/content_of_pact-f.html
Thursday, 20 June 2013 17:46
Working Group - a Portrait: PharmaInformatic Featured
InVitroJobs presents scientists and their innovative research in a regular feature called “Working Group – a Portrait”. We will focus on newly developed methods, their evaluation and their potential for reducing and where possible replacing animal experimentation according to the 3R principles of Russel & Burch (reduce, refine, replace). In this issue, we present PharmaInformatic from Emden, Germany.




 Dr. rer. nat.
Dr. rer. nat. Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.
Menschen für Tierrechte - Tierversuchsgegner Rheinland-Pfalz e.V.